
PURIHILO PN
3.2%-40mg(H-HA) + 40mg(L-HA) & PN
Main Composition
40mg of High molecule Hyaluronic Acid(H-HA) + 40mg of Low molecule Hyaluronic Acid(L-HA), PN (Polynucleotide)
Quantity
2.5 mL
Packing
1 Syringe / 1 Box
Country of Origin
REPUBLIC OF KOREA
Details about PURIHILO PN
AETERDERM®
PURIHILO PN
3.2 % - 40 mg (H-HA) + 40 mg (L-HA) / 2.5 mL
1.0 % - 25 mg (PN) / 2.5 mL
Hyaluronic acid sodium salt + Polynucleotide
1.0 % - 25 mg (PN) / 2.5 mL
Hyaluronic acid sodium salt + Polynucleotide
Mixing and Stabillizing H-HA & L-HA
ABEP™

HYBRID Technology
PCT International Patented


H+L & PN
HIGH-CONCENTRATED
HYDRATION & REVITALIZATION
HIGH-CONCENTRATED
HYDRATION & REVITALIZATION
HA concentration (80 mg / 2.5 mL) & Polynucleotide (25 mg / 2.5 mL)
H+L Synthesis & Stabilized
by Acid-base Equilibrium according to
by Acid-base Equilibrium according to
ABEP™

HYBRID Technology
PCT International Patented
Prevents HA denaturation by not using thermal synthesis
NO BDDE or other cross-linking agents used

Stabilized H+L Hyaluronic Acid Hybrid Complexs

H+L HA Complex
Highly Restore Hydration
Leading to a remodeling of the extracellular matrix in terms of
Elasticity
Support
Lifting Effect by Stable Skin Structure
Promoting and maintaining the viability of
Fibroblasts
Keratinocytes
Adipocytes
Polynucleotide DNA Polymer

PN
Rejuvenating & Revital Actions
via Activating 4 growth factors and Cytokine
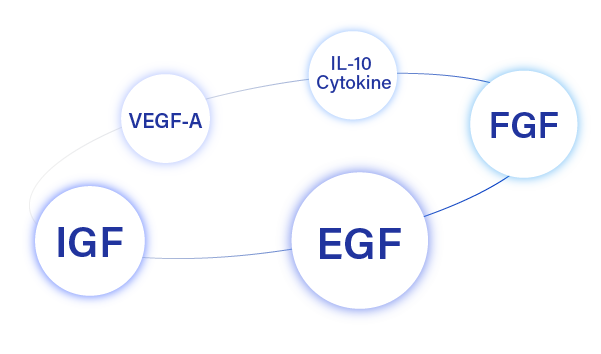
Stimulates Collagen & Elastine Synthesis
via Salvage pathway
Tissue remodeling and improvement in skin laxity
Face
Neck
Body
Hands
L-HA
40mg of Hyaluronic Acid
Low molecular weight
Low molecular weight
(200~400 kDa)
_graphic.webp)
H-HA
40mg of Hyaluronic Acid
High molecular weight
High molecular weight
(1300~1500 kDa)
_graphic.webp)
Starting with a
Simple Mix
Simple Mix
ABEP™
HYBRID Technology
PCT International Patent

The simple mix makes Hybrid complex by Acid-base equilibrium, according to ABEP process
(No chemical cross-linking agents used)
(No chemical cross-linking agents used)
PURIHILO's
H+L HA
Stabilized hybrid
cooperative complexes
cooperative complexes
Highly manageable
Low viscosity
Low inflammatory response

● Prevents HA denaturation by not using thermal synthesis
● High HA concentration
● NO BDDE or other chemical agents
● Stabilized hybrid HA with a duration comparable to a low cross-linked gel
PURIHILO's
PN(Polynucleotide)
contained in the
PURIHILO PN
PURIHILO PN
_optimized_DNA_graphic.webp)
● PURIHILO PN uses only certified medical grade Polynucleotide(PN) that have achived high results in CoA analysis.
● This polymers heals the skin from dermal layer, prevents aging, and revitalizes elasticity.
HYDRATION
PURIHILO PN is intended to provide DEEP SKIN HYDRATION,
will brings you a rejuvenated natural glow and revialized radiance.
will brings you a rejuvenated natural glow and revialized radiance.
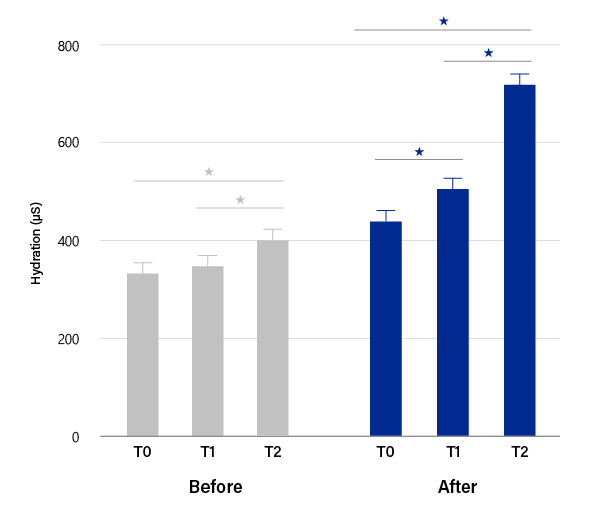
Facial skin hydration
mean ± SEM values (*P < .01).
mean ± SEM values (*P < .01).
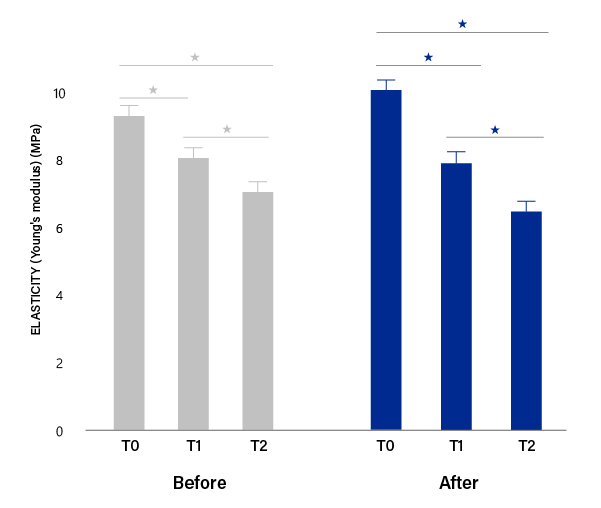
Facial skin elasticity (Young's modulus)
mean ± SEM values (*P < .01).
mean ± SEM values (*P < .01).
Ref. Carmen Laurino, MSc., Beniamino Palmieri, MD., & Allessandro Coacci, MD. (2015).
Efficacy, Safety, and Tolerance of a New injection technique for High- and Low-Molecular_Weight Hyaluronic Acid Hybrid Complexes
Efficacy, Safety, and Tolerance of a New injection technique for High- and Low-Molecular_Weight Hyaluronic Acid Hybrid Complexes
COLLAGEN SYNTHESIS
PURIHILO PN STIMULATES THE FORMATION OF COLLAGEN,
which in turn improves fine lines and wrinkles, producing firmer skin.
which in turn improves fine lines and wrinkles, producing firmer skin.
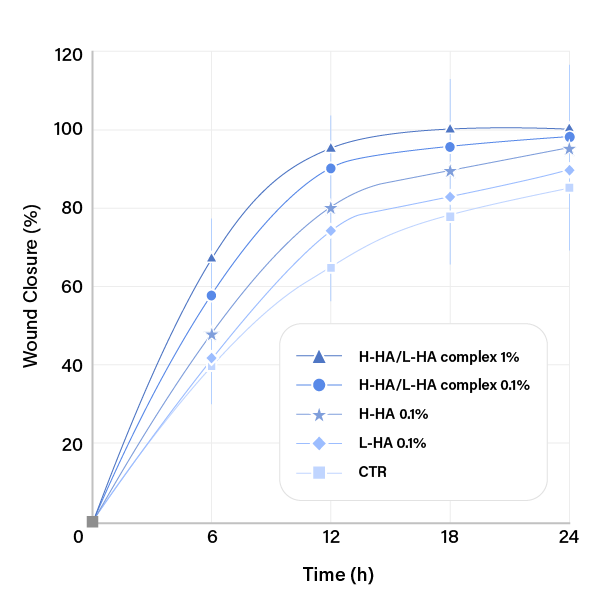
CONTROL
PURIHILO PN

UNTREAETED
HA 0.1%(w/v)
L-HA 0.1%(w/v)
H-HA/L-HA complex 0.1%(w/v)
H-HA/L-HA complex 1%(w/v)
Ref. D'Agostino et ai. BMC Cell Biology (2015) 16:19
MOISTURIZING EFFECT
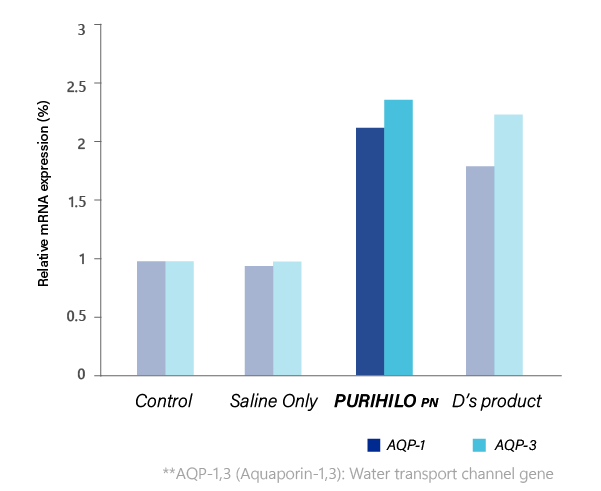
Each product was treated on the human skin dermal cells, and the genes expression level of the skin moisturizing factors were compared.
COLLAGEN SYNTHESIS EFFECT
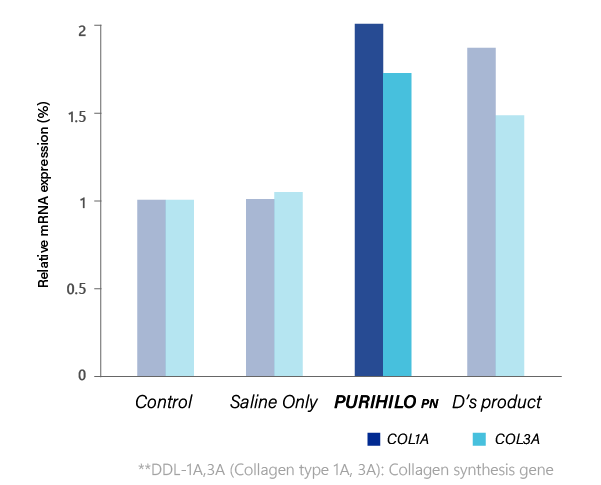
Each product was treated on the human skin dermal cells, and the genes expression level of the collagen synthesis factors were compared.
TISSUE REMODELING
PURIHILO PN is intended to REMODELING of the extracellular matrix, it will help you achieve a naturally youthful appearance as WRINKLES AND SAGGING SKIN REDUCTION.
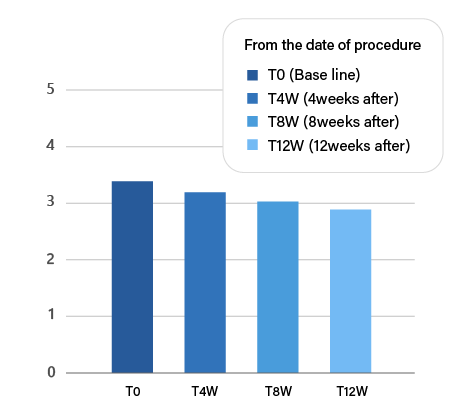
Reduction in the WSRS "wrinkle severity" throughout the study.
Note:*P<0.05vsT0. Abbreviation:WSRS, wrinkle severity rating scale.
Note:*P<0.05vsT0. Abbreviation:WSRS, wrinkle severity rating scale.
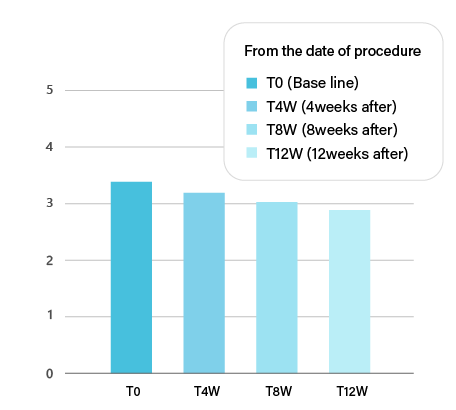
Reduction in the FVLS "facial volume" throughout the study.
Note:*P<0.05vsT0. Abbreviation:FVLS,facial volume loss scale.
Note:*P<0.05vsT0. Abbreviation:FVLS,facial volume loss scale.
TISSUE REPAIR
PURIHILO PN supports in the dermal TISSUE REPAIR process, in case of acne of scar.
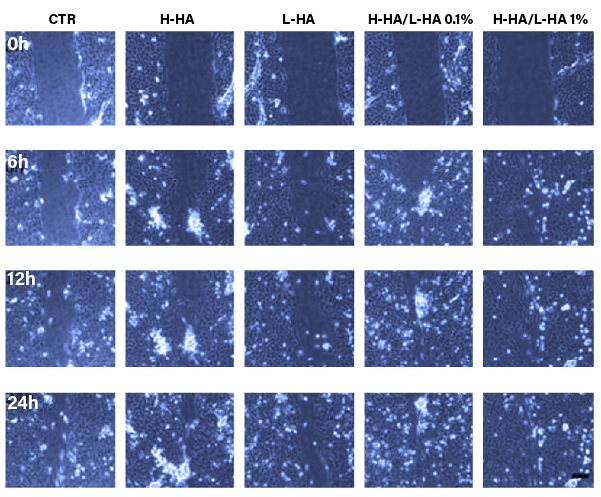
Representative micrographs pictures of HaCaT-HDF scratch assays immediately after the scratches, and in time course of the experiments.
Repair area percentage for the control and in presence of following stimuli.

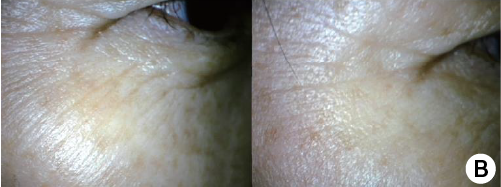
Around the eyes & crow’s feet
2 Sessions with a one month interval
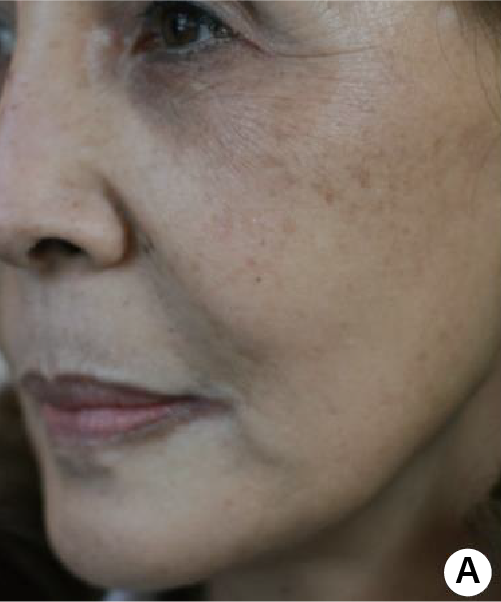
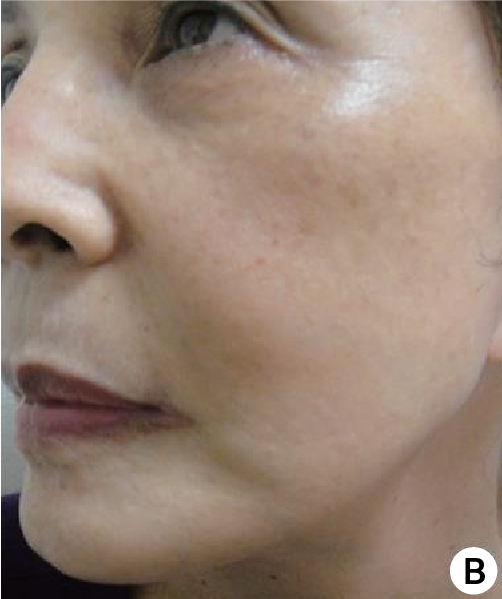
(A) Before treatment. The skin of patient looked dry and many wrinkles were seen especially crow's feet, nasolabial fold and vertical wrinkle on upper lip.
(B) The skin became healthier and tightened even with some whitening effect by skin revitalization.
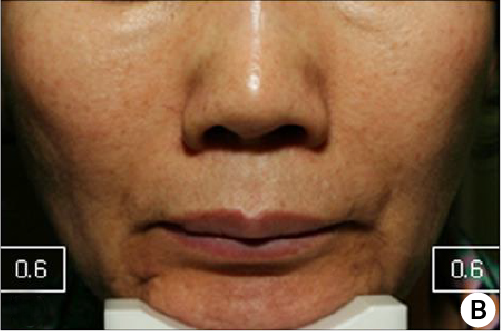
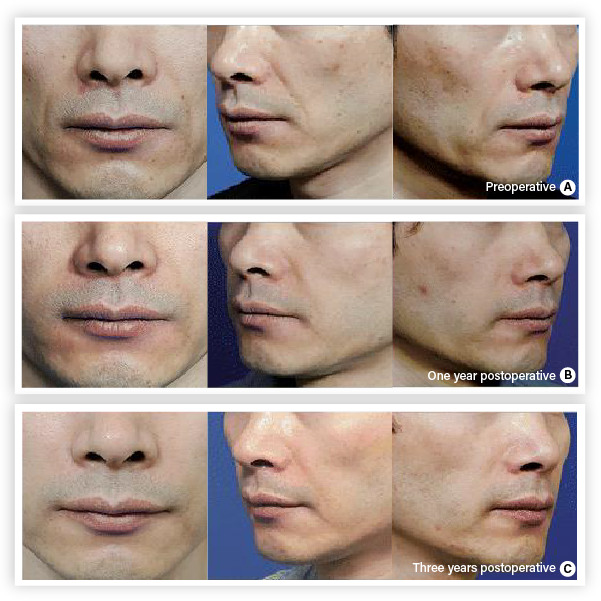
Subject(61 years/female) with PURIHILO PN in right nasolabial.
Baseline
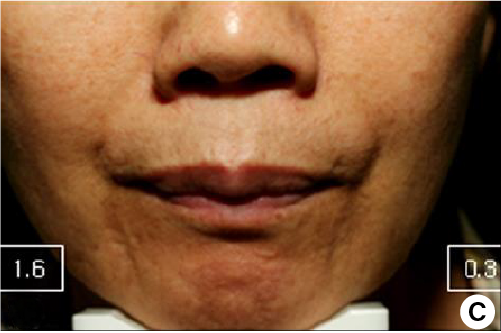
4 weeks after treatment
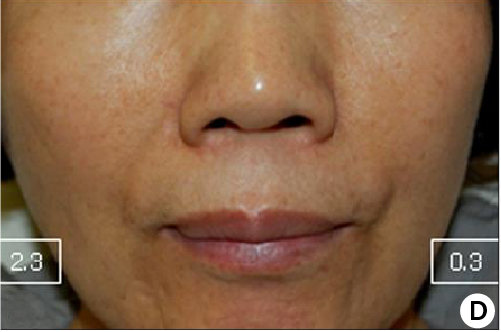
12 weeks after treatment
24 weeks after treatment
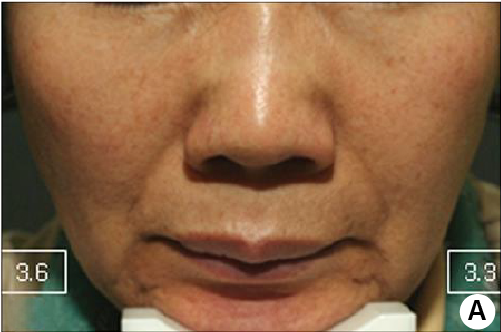
Nasolabial & around the eyes area
Before treatment and after 13 days. The amount of each shot was 0.001 cc (1 microliter). I administered 1,000 shots in a single session for the face, using a total of 1 cc (1 mL).
The crow's feet and tear trough were improved by dermal injection with mesogun.
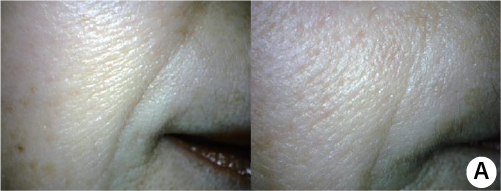
Focus on Nasolabial Folds


The nasolabial fold was improved also just by one session of schematic dermal injection using mesogun.
Coreection of deep sasolabial folds with PURIHILO PN.
(A) Preoperative view.
(B) One year after the injection
(C) Three years after the injection
(A) Preoperative view.
(B) One year after the injection
(C) Three years after the injection
You can use all aesthetic injection techniques
in the superficial subcutaneous layer.
First Session Results last up to 6 weeks, with approximately 50% of the effect remaining.
Second Session (6 weeks later) Extends results for an additional 3 months.
Third Session (3 months later) Prolongs the effects for up to 6 months.
According to
BAP TECHNIQUES (Bio Aesthetic Points)
is the technique for injecting to specific points anatomically receptive areas with an absence of large vessels and nerve branches and potential injury to vital structures, minimizing the risks while maximizing the diffusion of the product.
this method
Helps restore sagging skin, wrinkles, signs of aging.
Fewer injection points, fewer side effects for customers.
Maximizes effectiveness with less active ingredients.
Reduces recovery time and minimizes the area of bruising.
MALAR & SUB-MALAR areas remodeling
Identify the 5 BAP injection sites on each side of the face
Inject 0.2 mL per bolus at the superficial subcutaneous layer

Zygomatic protuberance
Make sure you are at least 2 cm away from the external side of the eye.
Make sure you are at least 2 cm away from the external side of the eye.

Nasal Base
- Draw a joining line between the nose wing and the tragus
- Draw a perpendicular line starting from the pupil midline
- Identify the injection point at the intersection point of the two lines
- Draw a joining line between the nose wing and the tragus
- Draw a perpendicular line starting from the pupil midline
- Identify the injection point at the intersection point of the two lines

Tragus
Make sure you are 1 cm in front of the lower margin of the tragus.
Make sure you are 1 cm in front of the lower margin of the tragus.

Chin
- Draw the midline of the chine
- Draw a perpendicular line one third from the top of the vertical line
- From the point of intersection move 1.5cm towards the oral commissures
- Draw the midline of the chine
- Draw a perpendicular line one third from the top of the vertical line
- From the point of intersection move 1.5cm towards the oral commissures

Mandibular angle
1 cm above the mandibular angle
1 cm above the mandibular angle
NECK areas remodeling
Identify the 10 BAP injection sites on the neck
Pinch the skin at the injection point
Inject 0.2 mL per bolus transversely across the skin at the superficial subcutaneous layer
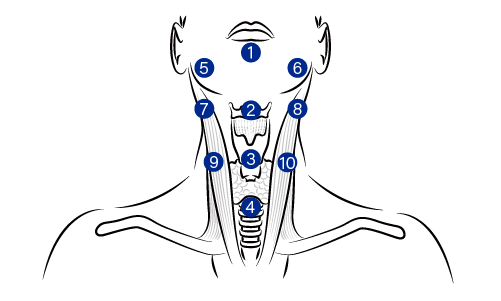
1 Midline : midway between the chin edge and hyoid bone
2 Midway between the Adam's apple and the thyroid cartilage
3 Midway between the hyoid cartiage and the manubrium
4 Manubrium apex
5 ~ 6 Under the mandibular angle at 0.5 cm from the medical edge of the SCM
7 ~ 8 On the horizontal axis of point 2 at 0.5 cm from the medial edge of the SCM
9 ~ 10 On the horizontal axis of point 3 at 0.5 cm from the medial edge of the SCM
NECK areas remodeling
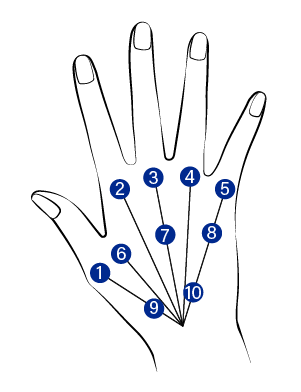
Identify the 10 BAP injection sites on the hand
Inject 0.1cc per point at distance of 1.5cm from the middle of the middle finger
3mm needle depth
Inject avoiding blood vessel locations