
LEXYFILL
H+L Hybrid Cross-linked HA Body filler
Main Composition
HA(hyaluronic Acid) 24mg / cc
Quantity
60 cc
Packing
1 Vial / 1 Box
Country of Origin
REPUBLIC OF KOREA
Suggested Collaboration
Details about LEXYFILL


Optimal Viscosity, Higher elasticity
Even without mixing saline, you can easily withdraw it from the vial

So there is no Volume-loss or deformation caused by saline. And it will show you amazing elasticity.
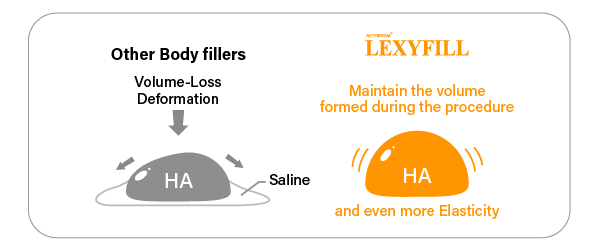
As a result, you can perform more sophisticated treatment.

We created body filler
with these amazing features
through long Cross-linking time
at low temperature
with these amazing features
through long Cross-linking time
at low temperature
For Accurate Body Styling
Other body fillers

Only use Large molecules of HA

A lot of BDDE was used, which can cause serious filler side effects.
Also these fillers are too hard to withdraw from the vial. So it should be combined with saline and shake it to mix
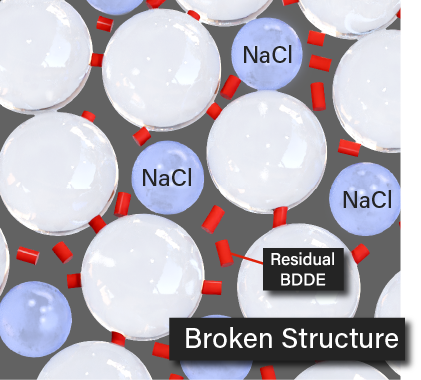
When you add the saline and shake it to mix, The HA cross-linking structure will be totaly broken. It cause incresing of residual BDDE and serious side effects and shock.
LEXYFILL body filler

Small molecules of HA were placed between it. So it has higher moisture and elasticity

Due to this HA structure, cross-linking can be done using very small amount of BDDE
So the probability of side effects is significantly reduced.

LEXYFILL can be easily drawn out with a syringe without mixing saline, so it can be injected with the HA structure intact.
Hyaluronic Acid

Biphasic crosslinked HA suitable for body volumizing
Repeat 14 times to remove 3 types of toxins.
Completely prevent side effects.
Completely prevent side effects.
◎ Residual BDDE
◎ Endotoxin
◎ Protein
◎ Endotoxin
◎ Protein
Maximize elasticity and retention by mixing large and small molecules of HA

HA Concentration
24 mg / mL
MOD(Degree of Modification, mol%)
24 mg / mL
Gel type
Biphasic
Storage Modulus(G')
100 - 250 Pa
Injection Force(N)
20 N
Viscosity & Elasticity test

References ) Sci Rep 2024:(5)

Breast Augmentation Procedure
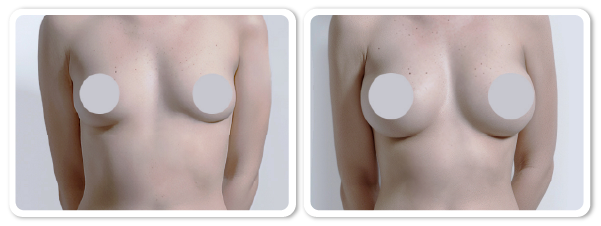
References ) Augmentation of breasts by VISIA-CR
Clinical trials by Dermatol Ther 2022:11:3-13
Clinical trials by Dermatol Ther 2022:11:3-13
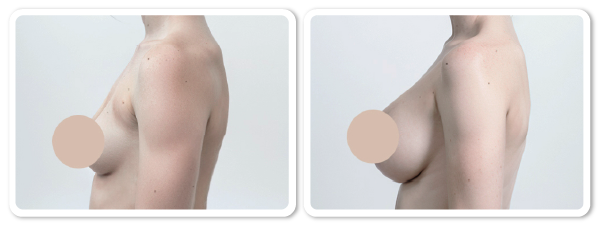
References ) Augmentation of breasts by VISIA-CR
Clinical trials by Dermatol Ther 2022:17:10-15
Clinical trials by Dermatol Ther 2022:17:10-15
Buttocks Augmentation Procedure
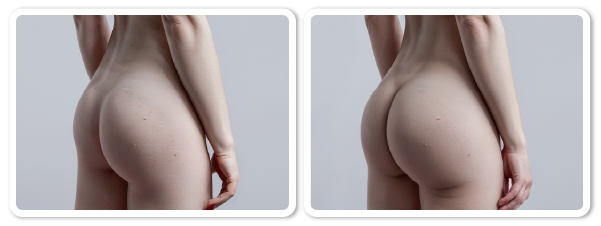
References ) Augmentation of buttocks by VISIA-CR
Clinical trials by Dermatol Ther 2022:21:35-19
Clinical trials by Dermatol Ther 2022:21:35-19
Treatment Area
Front Body 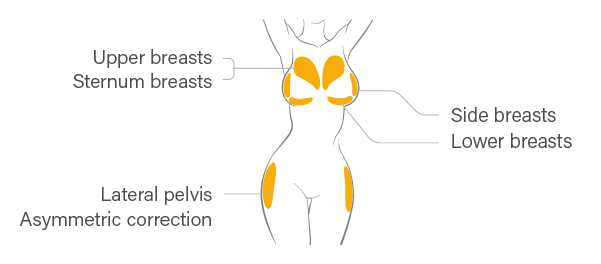
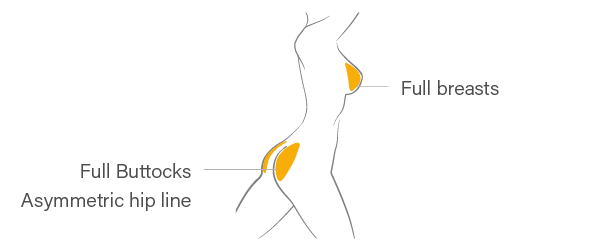
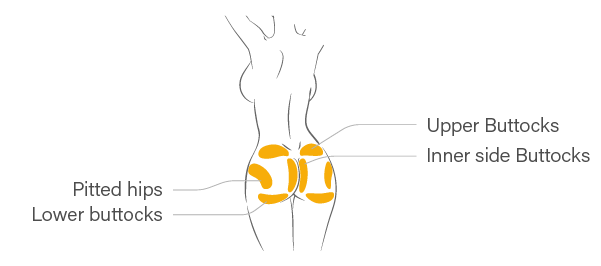
※ If patient have severe skin laxity or severe case of ptosis in the buttocks, it is strongly recommended not to inject hyaluronic acid filler into the lower part of the buttocks.
Typical Injection Site
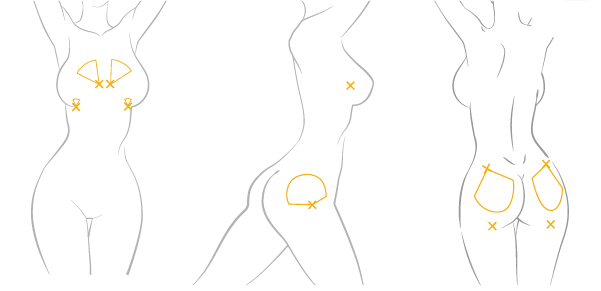
recommended Cannula
◎ Breasts augmentation - 14G 90mm micro-cannula recommended
◎ Upper breasts augmentation - 20G 70mm micro-cannula recommended
◎ Buttocks augmentation - 18G 70mm or 18G 90mm micro-cannula recommended
Treatment Process
◎ Check whether the patient is in a suitable condition for the procedure.
◎ Wash the treatment area with soap and water.
◎ Disinfect the treatment area with Alcohol or other antiseptic.
◎ Inject LEXYFILL HA body filler into the subcutaneous tissue plane of treatment area using thin-wall gauge needle or cannula.
◎ When injection is completed, treated site should be gently massaged so that it conforms to the contour of the surrounding tissues.
※ Detailed application instructions are provided in the manual enclosed with the product.
Precautions After Treatment
◎ Apply ice packs to the treated area for 15 to 20 minutes at a time, several times a day, to help reduce swelling and bruising.
◎ Avoid putting pressure on the treated area for at least 24 hours after procedure.
◎ Avoid for at least 48 hours after procedure
- Strenuous physical activity or exercise
- Alcohol and smoking
- Hot baths, saunas, and steam rooms
※ Detailed precautions are provided in the manual enclosed with the product.

